 Its units are the change in energy ( \Delta E \text. The equation for the change of entropy (delta S) of a system or object is the energy transferred to or from the object (Q), measured in Joules, divided by the average temperature of the object (T. This random motion, this entropy, is represented by S, and is a measure of the energy that is unable to do work. This law explains that concentrated energy has a tendency to become dispersed energy in the form of random thermal motion.ģ. This means that the entropy of a closed or isolated system will always increase over time. The second law states that the entropy of the universe is always increasing. It states that energy cannot be created or destroyed, only transferred between types of energy, such as heat and work.Ģ. According to this equation, entropy, symbolized by S, is equal to Boltzmanns constant, k, times the natural log of W, and W represents the number of microstates in a system. The second law of thermodynamics is a fundamental principle in physics that deals with the concept of entropy and the direction of natural processes.  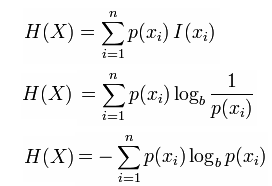
The first law of thermodynamics is the theory of conservation of energy. Finally, we will revisit the topic of Gibbs free energy, of which you should already have a decent understanding, and how it relates to enthalpy vs. Clausius discovered that, when a small amount of heat Q is supplied to a system, which is at an absolute temperature T, then it will undergo a process, and the. Once you have a firm hold on the definition of enthalpy, we will discuss entropy and look at entropy practice questions. Then we will talk about the definition of enthalpy and how to calculate it using enthalpy practice questions. This tutorial explains the theory behind enthalpy and entropy using the laws of thermodynamics. By substituting the general equation of state for any gas, pT(v)(v)F(v,T) in the thermodynamic equation, Tdsdu+pdv, and by applying the principle of. R., 2010, Advanced Heat and Mass Transfer, Global Digital Press, Columbia, MO.When you get to the thermodynamics section of AP® Chemistry, one of the hardest things to remember is the definition of enthalpy vs. The second law of thermodynamics requires that each of these entropy generations be greater than or equal to zero.įaghri, A., and Zhang, Y., 2006, Transport Phenomena in Multiphase Systems, Burlington, MA.įaghri, A., Zhang, Y., and Howell, J. The entropy generation for a control volume including Π phases consists of entropy generation in each phase, plus that in the interfaces. H -92.6kJ/mol Solution We can use the formula S 2 (NH3) (N2) + 3 (H2) S (2) (192.5 JK-1mol-1) 191.6JK-1mol-1 + (3) (130.6 JK-1mol-1) S -198. If the control volume includes Π phases, the second law of thermodynamics must be obtained by integrating over the two phases separately (Faghri and Zhang, 2006), (3), one obtains the integral form of the second law of thermodynamics for single phase systems: Introduction The macroscopic determination of entropy first was expressed by Clausius in 1865. Using the equation of state of the ideal gas, on the ends of the isotherm one has. The last term represents entropy generation, which should always be greater than or equal to zero, i.e.,Ĭombining eqs. Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. (2), the first term represents the change of entropy due to heat transfer across the boundary of the control volume, and the second term represents the change of entropy due to internal heat generation in the control volume. 
The change of entropy in a closed system results from heat transfer and/or entropy generation: If the state is a mixture of liquid and vapor, the entropy can be. bits, where is the probability that is in the state, and is defined as 0 if. The (Shannon) entropy of a variable is defined as. In mathematics, a more abstract definition is used. The entropy change for a system with fixed-mass and only one phase can be obtained by setting Φ = S,φ = s in eq. 3Nh2 32 + 5 from the microcanonical (NV E) ensemble. Entropy is a thermodynamic property, the value of entropy depends on the state of the system. In physics, the word entropy has important physical implications as the amount of 'disorder' of a system. The second law of thermodynamics requires that the entropy generation in a closed system (fixed-mass) must be greater than or equal to zero. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
